A Rat on a Cruise Ship Reminded Me That Viruses Do Not Read Specialty Lines
Pregnant patients are among the most vulnerable hosts for dozens of viruses
Three people dead on a cruise ship in the Atlantic. One confirmed hantavirus case. A virus carried by rodents, not typically associated with ships or obstetric care. Yet pregnant patients are among the most vulnerable hosts for dozens of viruses — and most of us were not trained to think systematically about all of them. This post is that list. obmd.com
Stylized image of hantavirus
Last week, while the MV Hondius was somewhere between St. Helena and Ascension Island, a 70-year-old Dutch passenger developed fever, headache, abdominal pain, and diarrhea. He died before reaching shore. His 69-year-old wife became ill on board and collapsed at O.R. Tambo International Airport in Johannesburg. She died there. A third passenger, a British national, tested positive for hantavirus. Three dead, six infected, one ship, one unusual pathogen.
The WHO is coordinating a multi-country response. The ship’s operator, Oceanwide Expeditions, has acknowledged the deaths while noting that hantavirus has not been definitively linked to all three. The investigation is ongoing.
Hantavirus is carried by rodents. It spreads when people inhale particles from dried droppings or urine. The Andes virus, found in South America, is the only strain known to spread person-to-person. The CDC has recorded 890 U.S. cases since surveillance began in 1993. This is not a common infection. Six cases on one ship is, in the words of a University of Michigan infectious disease specialist, something she had never heard of before.
I am not an infectious disease specialist. I am an obstetrician. But when I read this story, my first thought was not epidemiological. It was clinical.
My pregnant patients go on cruises. They travel to South America, sub-Saharan Africa, Southeast Asia. They clean mouse traps in their garage. They live in houses, not isolation chambers. And they carry a passenger whose immune relationship with the world has been fundamentally altered by pregnancy.
Pregnancy is not immunosuppression in the classical sense. It is immune modulation. The body performs a sophisticated tolerance trick to prevent rejection of a genetically foreign fetus, and that trick has consequences. Viral infections that cause mild illness in a non-pregnant adult can cause preterm labor, fetal loss, congenital anomalies, or maternal death in a pregnant patient. The virus does not change. The host does.
This is not a story about hantavirus specifically. It is a story about the full landscape of viruses that matter in obstetric care, and whether we are thinking about all of them every time we take a history, counsel a traveler, review immunization status, or manage a febrile patient in labor.
The answer, in my experience, is often no.
We are excellent at HIV. We screen universally, and our protocols for antiretroviral therapy and delivery management have reduced vertical transmission to under 1% in managed settings. We are good at hepatitis B because newborn immunization protocols and HBIG administration are embedded in delivery room practice. We think about HSV near term. We worry about varicella in the non-immune patient.
We are less systematic about the rest of the list.
CMV is the most common congenital infection in the world. It causes more permanent hearing loss and cognitive impairment in children than any other infectious agent. We do not screen for it universally in pregnancy. Most patients have never heard of it. Most of the public conversations about birth defects do not mention it. We counsel patients about cat litter and Listeria but not about the daycare worker sharing a cup of juice with a toddler who is shedding CMV in her saliva.
Parvovirus B19 causes hydrops fetalis. It is not rare. It circulates in schools and daycares. A school teacher or childcare provider who becomes infected in the second trimester faces the real possibility of fetal anemia severe enough to require intrauterine transfusion. The recognition window is narrow and the clinical picture in the mother is often unremarkable.
Zika is not gone. It is quieter. But the Aedes mosquito has not retired, and our patients still travel to endemic regions. Congenital Zika syndrome is one of the most devastating congenital infections documented in the modern era: microcephaly, cortical malformations, hearing loss, ocular defects. The counseling burden for a patient with a potential exposure and an ongoing pregnancy is substantial, and the evidence base for timing, testing, and surveillance is still not what it should be.
Hepatitis E deserves more attention in obstetric practice than it receives. In endemic regions, the case fatality rate for pregnant patients in the third trimester can exceed 25%. This is not a footnote in an ID textbook. It is a reason to take a careful travel and dietary history in every pregnant patient who presents with jaundice or hepatic dysfunction.
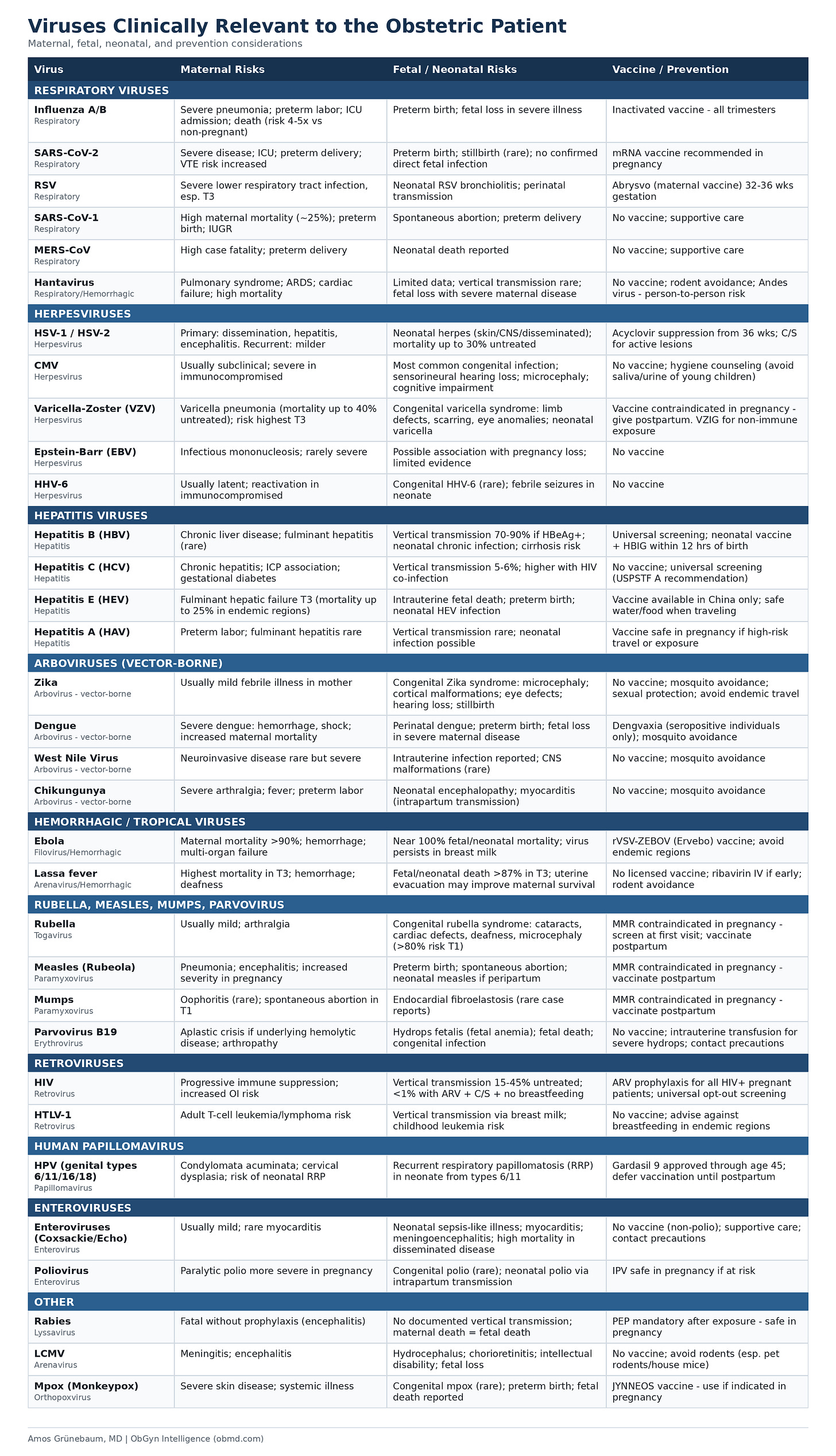
I have put together the table below as a working clinical reference. It is not exhaustive, and it is not a substitute for infectious disease consultation when you need one. But it is the list I wish I had seen as a resident. Every virus on it has documented maternal, fetal, or neonatal consequences. Every one of them has a potential interaction with pregnancy that changes management.
The table organizes them by category, summarizes the relevant maternal and fetal risks, and notes transmission routes and prevention strategies. I have included vaccines and interventions only where they are evidence-based, approved for use, or specifically recommended in or around pregnancy. I have not filled gaps with optimism.
A few observations from this table.
First, the vaccine column is shorter than it should be. We have effective vaccines against influenza, COVID-19, RSV, hepatitis A, hepatitis B, rubella, measles, mumps, varicella, HPV, polio, and rabies. Several of these are contraindicated during pregnancy (live vaccines) but should be given postpartum before the next conception. Others are specifically recommended in pregnancy. The prenatal visit is an immunization opportunity. We do not always use it as one.
Second, vertical transmission risk varies enormously within the table. For HIV without intervention, vertical transmission is 15 to 45 percent. For hepatitis B with HBeAg positivity and no prophylaxis, it approaches 90 percent. For CMV primary infection in the first trimester, the risk of congenital infection is approximately 30 to 40 percent, with a subset developing symptomatic disease. For rubella in the first trimester, the risk of congenital rubella syndrome exceeds 80 percent. These are not small numbers.
Third, the table contains several viruses that most practicing obstetricians will never encounter in clinical practice in the United States. LCMV, Lassa fever, Ebola. But global travel has made the world smaller. A patient who spent three weeks in West Africa visiting family, or who was on a cruise ship that stopped in South America, is a patient who belongs in front of an obstetrician with a complete viral differential. The geography of risk has changed. The training has not always kept pace.
The hantavirus cluster on the MV Hondius was unusual. But it reminded me of something that does not require a cruise ship or a rare outbreak to be true: the pregnant patient is a uniquely vulnerable host, and the viruses that threaten her are not limited to the ones on the standard prenatal panel.
My Take
The prenatal visit is one of the most intensive scheduled contacts between a patient and the healthcare system. We draw blood, check blood pressure, measure fundal height, review medications. We do not routinely ask about rodents.
That is not a criticism. You cannot ask about everything. But the viral risk landscape in pregnancy is broad, and the standard first prenatal visit template does not capture most of it. Travel history, occupational exposures, household contacts, immunization history, sexual history — these are the vectors that bring most of these viruses into the obstetric encounter. A teacher in a classroom full of young children is in a different CMV and parvovirus risk category than a remote worker who leaves the house twice a week. A patient who just returned from a Zika-endemic country is in a different conversation than one who spent her vacation in Maine.
We collect the information we are trained to collect. This table is an argument for expanding the frame.
The pregnant patient does not exist inside an infectious disease specialty line. Viruses do not check credentials at the door. The obstetrician who sees a febrile patient in the third trimester should be comfortable thinking across this entire table, not just the pathogens covered in a standard infectious disease in pregnancy lecture from 1995.
Three people died on a ship in the Atlantic from something carried by rodents, spread by breath, and diagnosed in a laboratory in South Africa while the vessel was off the coast of Cape Verde. It took a lot of geography to get to a simple conclusion: the world is smaller than our specialty silos.
If this post is useful, forward it to a colleague or a resident. If something on the table is wrong or missing, tell me. I update these references when the evidence changes.
Subscribe to ObGyn Intelligence for evidence-based commentary on women’s health every week — no sponsored content, no wellness industry partnerships, no hedging when the data is clear.
Selected References
1. Petri AE, Albeck-Ripka L. 3 Dead of Suspected Hantavirus Infections on Cruise Ship, W.H.O. Says. New York Times. May 3, 2026.
2. Centers for Disease Control and Prevention. Hantavirus Disease Cases Reported in the United States, by Year Reported, 1993-2023. Atlanta: CDC; 2024.
3. Silasi M, Cardenas I, Kwon JY, Racicot K, Aldo P, Mor G. Viral infections during pregnancy. Am J Reprod Immunol. 2015;73(3):199-213.
4. Swamy GK, Heine RP. Vaccinations for pregnant women. Obstet Gynecol. 2015;125(1):212-226.
5. Rawlinson WD, Boppana SB, Fowler KB, et al. Congenital cytomegalovirus infection in pregnancy and the neonate: consensus recommendations for prevention, diagnosis, and therapy. Lancet Infect Dis. 2017;17(6):e177-e188.
6. Mor G, Cardenas I. The immune system in pregnancy: a unique complexity. Am J Reprod Immunol. 2010;63(6):425-433.
7. Brasil P, Pereira JP Jr, Moreira ME, et al. Zika Virus Infection in Pregnant Women in Rio de Janeiro. N Engl J Med. 2016;375(24):2321-2334.
8. Khuroo MS, Kamili S. Aetiology, clinical course and outcome of sporadic acute viral hepatitis in pregnancy. J Viral Hepat. 2003;10(1):61-69.
9. CDC. Recommended Adult Immunization Schedule for Ages 19 Years or Older, United States, 2024. MMWR Recomm Rep. 2024.
10. WHO. Hantavirus Disease — Multi-country Event. Disease Outbreak News. May 4, 2026.